Elmiron lawsuits claim Janssen Pharmaceuticals was aware of the risk of eye damage but did not provide adequate warnings. The manufacturer also failed to disclose the risks to the US Food and Drug Administration and the medical community.
If you or your loved one developed vision or eye damage after using Elmiron, you might qualify to file a lawsuit against Janssen.
Join the Many can help you pursue financial compensation to cover treatments and damages for your pain and suffering.
A Brief History of Elmiron

Elmiron is a semi-synthetic polysulfated xylan. It’s a type of weak blood thinner but functions by protecting the bladder while treating conditions such as interstitial cystitis (IC).
Elmiron comes from Janssen Pharmaceuticals, a branch of Johnson & Johnson. The FDA first approved Elmiron in 1996, becoming popular in the medical community for treating IC.
Elmiron is still the only FDA-approved oral drug for treating bladder pain related to interstitial cystitis. It’s also effective against bladder pain caused by urinary tract infections.
While Elmiron provided a solution for IC, research shows it also caused severe side effects such as vision issues and eye damage.
Concerns about the adverse side effects of prolonged use of Elmiron first came into the limelight in 2018. Doctors noticed that some people who used the drug for five or more years experienced a loss of eyesight, vision issues, and retinal damage.
These findings raised concerns because thousands of patients relied on Elmiron to treat IC. These patients took the drug for years, unaware of the high risk of eye damage or vision loss.
The discovery sparked lawsuits against Janssen for failure to provide enough warnings on the side effects.
There’s conclusive evidence connecting Elmiron to retinal damages for product liability lawsuits. If you used Elmiron and developed retinal issues, you may have a valid case to hold Janssen Pharmaceuticals liable for your injuries.
Overview of Research Findings

Interstitial cystitis is a challenge for most people, with up to 12 million people in the US suffering from IC. This bladder condition increases urination frequency, and patients experience discomfort and chronic pain.
Elmiron is effective against symptoms of IC because it prevents the bladder walls from swelling and irritation. It works like a synthetic mucus layer, forming around the bladder to mitigate irritation.
Since 1996 when the drug entered the market, doctors have considered Elmiron a relatively safe drug with few severe side effects or risks. This changed after the researchers discovered a possible link between Elmiron and retinal damage in 2018.
Doctors from the Emory Eye Center in Atlanta, Georgia, discovered a trend in some patients. The patients had pigmentary maculopathy and unusual damage to the macula region.
They developed these conditions after prolonged use of Elmiron.
The Emory Eye Center published a preliminary report after the discovery, warning that the drug could be toxic to the human eye’s retinal tissue. After the report, ophthalmologists from Northern California studied Elmiron’s safety and toxicity.
The team reviewed thousands of patient records with a history of using Elmiron. They presented the findings at the annual American Academy of Ophthalmology meeting in October 2019.
The alarming study results indicated that Elmiron was highly toxic to sensitive retinal tissue. From the patient records, nearly 25% of patients using Elmiron had significant eye damage or early symptoms of maculopathy.
The doctors tested ten patients, half undergoing fundus examinations and the other half imaging. On average, the ten patients took Elmiron at a mean total dosage of 380g for about 4.2 years.
The researchers found clear indications of drug toxicity due to prolonged use. The imaging tests detected Elmiron-related maculopathy. Most of the patients already experience total or partial loss of their vision.
What Eye Conditions Does Elmiron Cause?
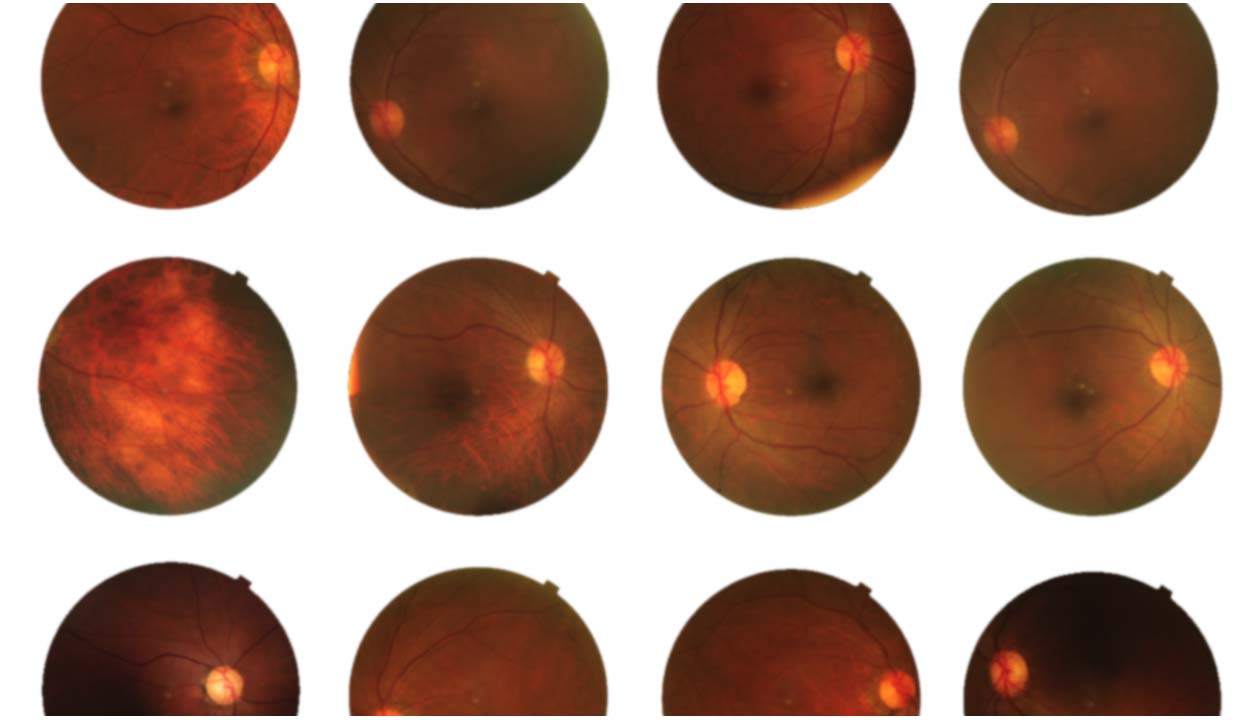
Several studies link Elmiron to a unique maculopathy known as pigmentary maculopathy. This type only affects patients who use Elmiron for an extended period.
The active ingredient in Elmiron is the only substance that causes pigmentary maculopathy.
Maculopathy is a general term for any pathologic condition or disease that affects the macula, which is the central part of the retina in the eye. Maculopathy is progressive, has no cure, and could cause blindness or total vision loss.
The 2019 study discovered that patients who took higher doses of Elmiron for longer had the most significant eye damage. The damage will likely continue advancing even after the patients stop taking Elmiron.
Common symptoms of Elmiron eye injuries are:
- Total vision loss
- Dark spots on or around the retina (Hyperpigmentation)
- Blurred vision
- Changes in the eye color
- Lack of light sensitivity or dim vision
- Blind spots in your vision
- Eye pain
- Challenges seeing close objects
- Muted or less vivid colors
- Dark spots in the center of your vision
- Challenges adjusting to the darkness
- Increased light sensitivity
- Difficulty seeing distant objects or reading
- Straight lines appearing squiggly or curvy
- Abnormal blood vessels around the macula
- Vitelliform deposits under the macula, which look like yellow lesions
If you are experiencing such symptoms, there’s a great chance you qualify for an Elmiron lawsuit.
The Elmiron Warning Label Updates

The International Cystitis Network (ICN) presented a Citizen Petition to the FDA in April 2019.
The petition required Janssen Pharmaceuticals to amend Elmiron’s product label and add warnings about the risk of eye damage. It also suggested including yearly eye examinations.
The European Medicines Agency (EMA) also demanded similar label changes in 2019, and Health Canada did the same in October of the same year. Janssen Pharmaceuticals complied with the recommendations and updated the warning label.
Meanwhile, Elmiron users in America still had no idea since there were no US updates then.
In June 2020, Janssen Pharmaceuticals finally updated Elmiron’s product label in the US. The current label warns that Elmiron can interfere with the pigments in the retina, causing issues such as:
- Sensitivity to low light
- Blurred vision
- Reading problems
The label warns that the symptoms could worsen even if you stop taking Elmiron, and the changes may be irreversible. Anyone taking Elmiron should have regular eyesight tests while continuing treatment. The first test should be within the first six months of starting treatment.
According to the label, this warning primarily applies to people who took Elmiron for three years or more. However, it also acknowledges cases where affected patients took the drug for a shorter period.
Although this additional information is helpful, it came after thousands of people had already used Elmiron for years. It makes no difference to anyone who already suffers from permanent eye damage.
Join The Many believes negligent corporations should be liable for their actions or failure to act. Reach out to discuss your Elmiron case and discover your legal options at absolutely no cost.
Lawsuit History

Affected patients began filing Elmiron lawsuits against Janssen Pharmaceuticals in 2020. The initial cases included claims from Kimberly Pelczar, Valarie Hull, and Jeanette Milburn.
According to the lawsuits, the three women used Elmiron for an extended period, causing them to develop the following:
- Retinal hemorrhage
- Vision degradation
- Loss of night vision
- Retinal injury
- Pigmentary maculopathy
They claimed Janssen Pharmaceuticals did not provide adequate warnings about their defective product. The failure to warn the women or their doctors posed a risk of permanent damage to their vision.
There’s a pending Elmiron class-action lawsuit against Janssen Pharmaceuticals filed in May 2020. The lawsuit claims the manufacturer did not warn them of the risk of vision loss and permanent eye damage.
The class action seeks compensation for damages such as legal fees and medical expenses.
Since Elmiron is a widely-used drug, courts continue to receive thousands of Elmiron lawsuits. The US Judicial Panel on Multidistrict Litigation (JPML) merged these lawsuits into a multi-district litigation (MDL) in 2020.
An MDL is different from a class-action lawsuit. In a class action, many people unite to file one big case and share the settlement amount if the case succeeds.
In an MDL, the cases remain independent, and the consolidation is to fasten the process and streamline discovery. The settlement amounts depend on individual matters.
In 2020, civil cases from courts across the country moved to one federal court in New Jersey. The Elmiron MDL (MDL 2973) is under Judge Brian Martinotti, a respected and experienced mass tort litigation judge.
Examples of Elmiron Pigmentary Maculopathy Lawsuits
In October 2021, Deborah Quick filed an Elmiron Pigmentary Maculopathy Lawsuit in the US District Court in Oregon. According to the lawsuit, Quick received the first Elmiron prescription in 2002 to treat IC. She continuously used Elmiron daily, per the prescription, from 2002 to March 2020.
This prolonged use caused Quick to start experiencing issues with her vision, which deteriorated with time. She was unaware the vision problems were due to Elmiron until her doctor notified her of the possible link in 2019.
Janssen Pharmaceuticals’ failure to warn caused her pigmentary maculopathy diagnosis. Quick’s lawsuit is part of the Elmiron MDL.
Joanne Williams’ Elmiron Lawsuit
Joanne Williams is a 79-year-old woman from Plymouth County, Massachusetts. Joanne got an Elmiron prescription from her doctor to manage the pain caused by her interstitial cystitis.
She took Elmiron between January and May 2015 and developed eye problems that led to vision loss. The drug caused retinal pigmentary degeneration, which was otherwise avoidable.
Joanne’s lawsuit seeks to hold Janssen Pharmaceuticals liable for her vision injury and diminished quality of life. She claims Janssen sold Elmiron to her and other healthcare providers while aware of the possible risks it carries.
The company failed to update the product, instructions, warnings, and packaging, leading her to believe Elmiron was safe.
Unsurprisingly, her attorney reached out to Janssen to potentially discuss an out-of-court settlement. The company never responded to the attorney or reached out to Ms. Williams.
By September 2021, the MDL had about 497 Elmiron cases and over 700 in February 2022.
So far, we’ve not seen any Elmiron lawsuit trials, but Elmiron attorneys are busy getting ready. The first bellwether trial in the MDL class action will start in January 2023.
Bellwether trials are test trials that determine how the following cases will proceed. The settlements set the stage for how valuation will happen and the expected timelines of a global compensation.
By June 2022, there were 1,360 lawsuits in the federal court and 1,740 claims in the MDL by September.
Do I Qualify to File an Elmiron Lawsuit?
You might have a valid case if you received a confirmed diagnosis of eye or vision problems after using Elmiron for an extended period. Most Elmiron attorneys use these guidelines to determine whether your case qualifies:
- You used Elmiron for about two years
- You received medical treatment for vision problems
- You got macular degeneration, retinal maculopathy, or pigmentary maculopathy after using Elmiron
- You suffered issues like vision loss, difficulty reading and adjusting to darkness after using Elmiron
- Your vision issues occurred when still using Elmiron or within twelve months after discontinuing use
- Your vision issues started on or after January 1, 2010
What Compensation Can I Expect From an Elmiron Lawsuit?

An Elmiron lawsuit is a product liability case under personal injury claims. In most product liability cases, the damages include:
- Punitive damages if the company is grossly negligent or acted maliciously
- Pain and suffering
- Past medical expenses and future medical bills
- Emotional distress
- Loss of wages and earning capabilities
- Loss of enjoyment of life due to illness and loss of vision
The particular settlement amount in your case will likely depend on the extent of your eye damage. For instance, if you have total vision loss, you’ll likely receive more than someone with increased light sensitivity.
It’s Time to Take Action; Join the Many.
If you or a loved one experienced eye or vision problems after using Elmiron, filing a lawsuit gives you the best chance to hold Janssen liable.
You deserve fair compensation to help cover your medical expenses, lost wages, and lower quality of life. Join the Many will guide you through the lawsuit process and connect you with the best attorneys to fight the case on your behalf. You’re not alone; we’re here to fight for people like you.