Did your child have the NuVasive MAGEC System placed? Is he or she struggling with pain and discomfort? Perhaps your child has even experienced the need for replacement procedures. This spinal treatment device has been used in many children, and some of them are suffering from significant complications from them.
Join the Many is working to support victims of NuVasive MAGEC System complications. Filing a lawsuit against the companies responsible for your suffering doesn’t have to be difficult; we can shoulder the burden so you can focus on living your life.
What Is the NuVasive MAGEC System?
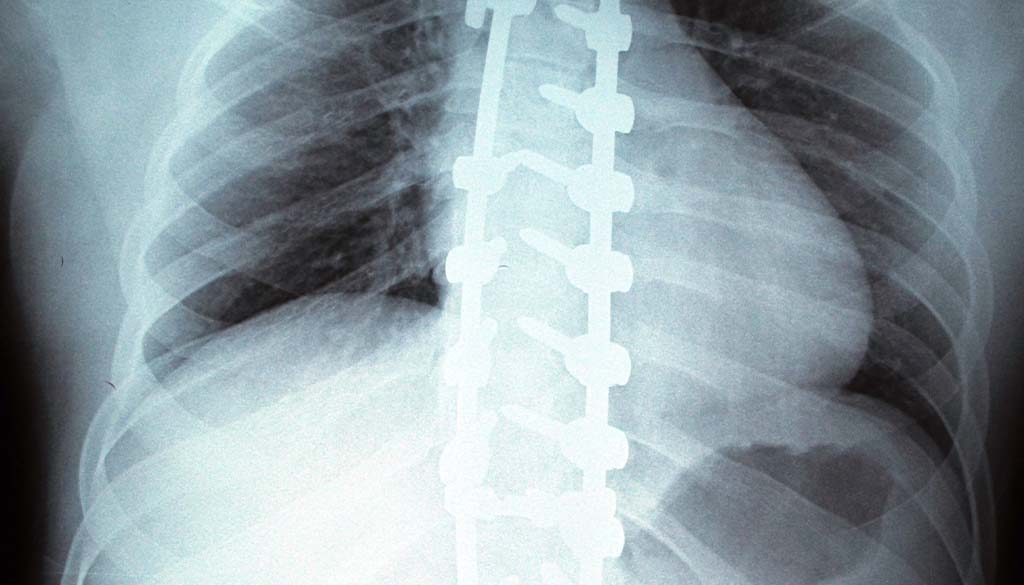
The NuVasive MAGEC System claims to be an innovative device that uses magnetic technology to improve the curvature of the spine in children who have scoliosis. The device uses growing rods that are placed within the spine of a child with the goal of enabling a straightening of their spine over a period of time. The device components are designed to help those with severe spinal deformities, according to the Children’s Hospital of Philadelphia.
The device includes magnetic rods that are positioned in the spine that can be extended over time remotely. They work as a type of brace for the spine while the child is growing, helping to minimize any worsening of scoliosis in them.
To understand why this product is novel, it’s important to know what the alternative treatments for this condition are. In some situations, the spine is fused, which prevents the worsening of the condition but also creates significant limitations to movement and overall growth. The alternative types of devices are effective as well, but they require a person to have multiple surgeries, sometimes as frequently as every 6 to 12 months, to adjust the rods as a person grows. That creates its own set of risks.
The NuVasive MAGEC System is designed to be placed for two years. During that time, doctors can adjust the rods using an external remote device. This prevents the individual from having to have multiple surgeries.
Surgeons claim that this product can help to improve a person’s quality of life and reduce risk, according to Seattle Children’s Hospital. This may be true for some people. However, there are risks to using the MAGEC System, and those are not always well understood by caregivers or users.
The MAGEC System is designed for:
- Early onset scoliosis (EOS)
- Treatment in those who are under the age of 10
- Treatment for conditions in which there are noticeable curves or twists
The U.S. Food and Drug Administration first approved the use of the NuVasive MAGEC System in 2017. This is a newer product and has not been approved for use in all situations. It is only used as a treatment for both severe and progressive EOS. It is only to be used when other methods that do not involve surgery have failed to be effective.
The device is implanted during a surgical procedure. They are left in for a maximum period of 2 years at a time. The rods are positioned on either side of the spine. When the child grows, the rods are then able to be lengthened, usually once every six months. This enables the growing rods to continue to support the patient’s needs. This adjustment can be done in a doctor’s office rather than in a surgery.
History of The NuVasive MAGEC System
The MAGEC System from NuVasive is the only such product like it. It is also a very new product that has only been approved for use in the U.S. since 2017. It is also approved in Europe.
The company working to produce these products completed various studies to see results. In studies conducted by the manufacturer and developers, it was noted that the device saw an improvement of between 14 and 17% with the use of the MAGEC treatment in patients with EOS.
While this is the case, it is also important to point out that during these studies, there was a high complication rate of 18%. The complications seen during testing involved wound infections, fixation failure, rod breakage, and prominent metalware, according to studies published in the National Library of Medicine.
Names of the NuVasive MAGEC System
- MAGEC System
- NuVasive MAGEC
- MAGEC growing rods
Overview of Research About the NuVasive MAGEC System
The NuVasive MAGEC System is the first of its kind, and that means that there is a lot of risk involved in the use of these products. While many children saw improvement using the device, there are risks to consider. More so, those with EOS may not see the results they hoped for.
After the product’s launching, numerous children had the device implanted in the U.S. Many of these children did well and did not have complications. However, the FDA has reported some concerns with the product – and it is critical for families to understand what those risks are.
In July of 2021, the FDA issued a notice to provide information about these devices to patients, caregivers of those patients, and health care providers. Specifically, the FDA alerted to potential mechanical failures of the product as well as risks associated with tissue incompatibility, also known as biocompatibility.
The FDA notice included various components of the NuVasive Specialized Orthopedics’ MAGEC devices, including:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X device
- MAGEC System Model X rod
- MAGEC System Rods
The concerns with the device are worrisome – and they can be hard to understand for those who are just getting them installed.
To be clear, the FDA did not remove the product from the market – they are still available for implant, and the FDA has said that the risks are outweighed by the benefits that the system offers to patients.
What Are the Problems?
A number of potential problems have occurred with the MAGEC System. Some of the complications related to the product include:
- Rod breakage
- Fixation failures
- Metal particles that break off from wear and embed in tissues
- Fractured drive pin
- Infection risks
- Autofusion of the spine
- Spinal deformities
- Screw breakage
- Tissue death
One of the main concerns relates to the endcaps located on the product. These sit over the top of the ends of the rods. In numerous situations, the endcaps can detach and cause numerous health challenges, including significant pain and infection risk.
Studies Show the Risks
A study published in the National Library of Medicine provided a look at the complications associated with the device. The study found that, in the use of this product, a full 48% of patients using it suffered some type of complications from use. That’s nearly half of all children who had the device implanted. They also found that 44% of those with these devices needed unplanned surgeries as a result of those complications, putting them even further at risk.
Recalls of the NuVasive MAGEC System
It is quite important for patients and caregivers to understand that these devices have been recalled – however, changes have been made.
In February 2020, the manufacturer, NuVasive, issued an Urgent Field Safety Notice for the MAGEC system. In that notice, they recalled the device components that were a known problem. This involved cases in which the endcap on the device separated from the actual rod, according to the FDA.
In July 2020, the FDA expedited and cleared a modified version of the MAGEC Model X rod. It approved the use of this new product as a way to potentially reduce risks and complications associated with its use.
In December 2020, the FDA received information about another Field Safety Notice issued by the manufacturer directed at the European Union. This notice alerted to risks associated with the biocompatibility of the product. The FDA then notes that it started receiving the same types of concerns from patients in the U.S. in early 2021. In this situation, patients described local tissue reactions related to the endcap separation events with the devices.
The FDA has not recalled any additional products. It also provided recommendations for those who had the device:
- If the patient is not experiencing complications, no treatment is recommended.
- It does not recommend the removal of the system unless complications warrant it.
- Those who are considering the use of the device should continue to do so because it offers benefits that are worth the risks in some situations.
- Individuals must be informed about all risks before this product can be used.
Lawsuit History of the NuVasive MAGEC System
For those who have suffered significant loss from the use of the NuVasive MAGEC System, such as surgical procedures, medical bills, infections, loss of mobility, and even loss of life, it is critical to reach out to learn about lawsuits that you could be a part of or how to file your own.
Right now, many attorneys are working on lawsuits related to the NuVasive MAGEC System. This is a very new situation, though, and not many of those cases have made it to a court of law just yet. These cases may end up coming to court in the coming months to years as more people come forward.
At this point, they are being managed on an individual basis by the local U.S. District Courts. The claims range widely and include a range of risks associated with the use of the NuVasive MAGEC System.
The manufacturer has not issued a statement regarding these products just yet. As a result of this, it is important to note that there are no settlements pending regarding the case.
Some of the claims made by these lawsuits include losses associated with the following:
- Cancer
- Bone abnormalities
- Pain and irritation
- Soft tissue inflammation
- Tissue damage or discoloration
- Complications from surgeries
Can You File a Claim Against NuVasive MAGEC System?
Yes, however, this is a new product, and the lawsuits are just starting against the company. That means that it is best to work with an attorney that is familiar with the MAGEC system as well as the losses others have suffered as a result of its use.
Join the Many can help you.
If your child used the NuVasive MAGEC System and experienced serious complications from the device’s use, including hospitalizations, surgical procedures that were unexpected, infections, and other complications, Join the Many.
We help you determine if you may have a legal case against the manufacturer. If you have a qualifying case, we’ll set to work handling every aspect of filing a claim, including connecting you with the best legal care for your situation.

