IV Flush Syringes often have pre-filled saline that helps to flush out medical devices that deliver drugs to the bloodstream via a catheter or needle.
When used and manufactured correctly, IV flush syringes are vital to patients and healthcare providers. These medical devices minimize the risk of infection and enhance workflow efficiency in healthcare settings. They also reduce the risk and frequency of needlestick injuries.
However, IV flush syringes carry a high risk of infection. If there’s bacterial contamination, the pathogens enter the bloodstream directly, which makes the condition lethal.
In 2016, the CDC became aware of a Burkholderia cepacia (B. cepacia) outbreak affecting five states. The agency investigated 162 cases of B. cepacia infections linked to contaminated Nurse Assist IV Flush syringes.
B. cepacia bacterial infection affects people who already have a weak immune system. People with existing lung conditions, pneumonia, and cystic fibrosis have challenges recovering from the initial illness.
If you or a loved one developed a bacteremia blood infection or sepsis after using a contaminated Flush IV syringe, you might qualify for a lawsuit. Join the Many support people like you with legal resources that take the hard work of building and fighting a case off your shoulders.
History of IV Flush Syringes
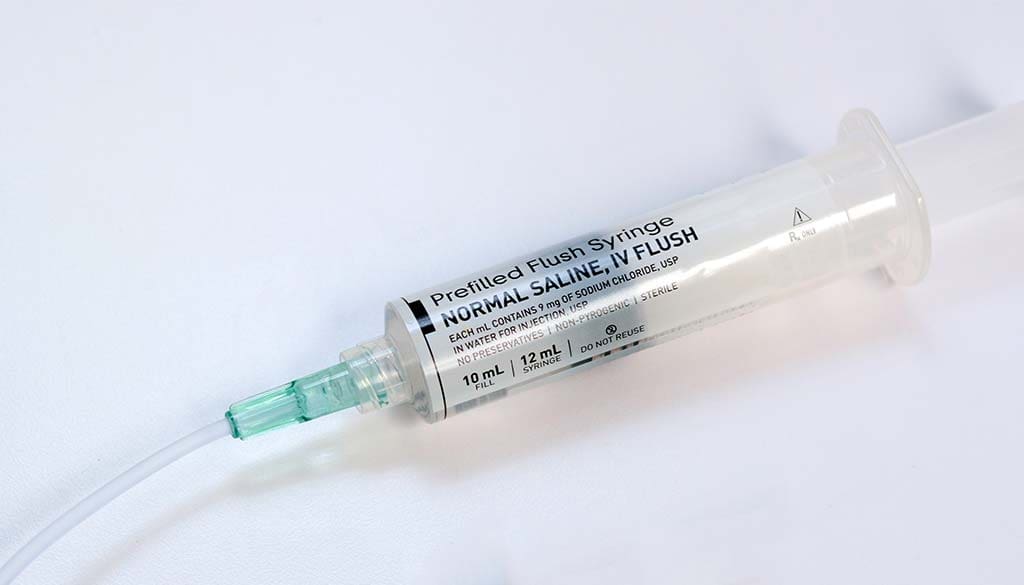
IV Flush Syringes are standard in various healthcare settings. Medical professionals often use these devices in various aspects of patient care, including:
- Surgery
- Pain management and anesthesia
- Pharmacy
- Hematology and oncology
- Home infusion therapy
- Emergency medicine
All medical devices should be effective and safe and comply with stringent quality measures.
However, some manufacturers fail to ensure their products are safe and contamination-free. They distribute defective devices that often cause severe injuries to unsuspecting patients.
In 2016, the CDC announced an investigation into contaminated syringe produced by Nurse Assist. This decision followed reports of B. cepacia bloodstream infection outbreaks caused by the needles.
Health officials identified the bacterial infection in 164 patients from 59 medical facilities across five states. The cases were in Maryland, Delaware, Pennsylvania, New Jersey, and New York.
Seven of the 164 patients lost their lives, but the CDC was unsure whether it was due to underlying conditions or the infection.
Most bloodstream infection cases were in New Jersey, where officials confirmed 59 cases. Health officials confirmed 58 cases in New York and 30 in Pennsylvania. Twelve cases were in Maryland, and the remaining 4 cases were in Delaware.
IV Flush Contamination
Luckily, the CDC discovered that Nurse Assist syringes were likely causing the infections. The investigators successfully controlled the outbreak, and the national recall of the contaminated needles prevented more infections.
Although it’s unclear how sterile IV flush syringes picked up the bacteria, the CDC shared some insights on how the contamination likely occurred.
According to Dr. Richard B. Brooks, a former CDC researcher, proper sterilization using gamma radiation was enough to kill the bacteria. The contamination likely occurred because the syringes did not undergo adequate sterilization.
The sterilization process also likely had issues that made it ineffective. Another possible explanation was that the saline inside the syringes came into contact with the bacteria after sterilization.
B. Braun’s Bacteria-contaminated Syringes / Contaminated IV Syringes
B. Braun Medical Inc. also came under scrutiny in 2016 after the federal government filed a lawsuit against the company. The case accused B. Braun of distributing contaminated pre-filled IV flush syringes in 2007.
In 2006, B. Braun contracted AM2PAT Inc. (previously Sierra Pre-Filled) to produce pre-filled saline syringes. B. Braun got the IV flush syringes from AM2PAT’s North Carolina facility and distributed them to healthcare facilities across the country.
During negotiations, B. Braun became aware of some manufacturing issues at the production facility. AM2PAT agreed to relocate the manufacturing plant and contract a different syringe sterilization company.
However, B. Braun resumed distribution of the syringes after the changes before their quality team had a chance to review the new sterilization process. They also did not visit the new facility for inspection.
B. Braun knew about the disputable manufacturing processes yet continued distributing the saline syringes. The contaminated IV flush syringes eventually ended up in health facilities nationwide.
Due to B. Braun’s negligence, the contaminated syringes got recalled within two months of starting distribution.
The first IV flush syringe recall occurred after discovering that the sterilization process had significant flaws. The process left some toxic impurities in the saline inside the syringes.
B. Braun continued selling the IV flush syringes, although AM2PAT had yet to review the sterilization procedures.
Shortly after, dozens of patients in different facilities in multiple states fell ill due to the bacteria-contaminated IV flush syringes. The patients reported experiencing symptoms such as:
- Nausea
- Vomiting
- Chills
- Infections
- Fever
- Sepsis
Patients who had weak immune systems suffered more complications. The Serratia marcescens bacterial infection caused life-threatening injuries and severe health complications.
The contaminated syringes sold by B. Braun caused bacterial infections in over 100 patients and at least five fatalities.
B. Braun eventually terminated the contract with AM2PAT in 2008 as the nationwide recall of the pre-filled IV flush syringes continued. Although the recall helped to prevent further infections, hundreds of vulnerable patients already suffered life-changing injuries.
Such negligent manufacturers should take responsibility for their actions or inaction.
If you believe you have a valid IV flush syringe lawsuit, Join the Many offers to connect you with the best legal team to fight on your behalf.
All Names IV Flush Syringes Go by
IV flush syringes contain Sodium Chloride at a concentration of 0.9 %. The generic name for these syringes is Normal Saline Flush. Various corporations manufacture(d) IV flush syringes in the US, including:
- Nurse Assist Inc.
- Hospira Inc.
- Becton Dickinson (BD) & Company
- B. Braun Medical Inc.
- Covidien LLC
- Medline Industries Inc.
- Excelsior Medical Corp
Some popular brand name examples include:
- HepMed IV Pack
- Swabflush
- BD PosiFlush SF
- BD PosiFlush
- Syrex
- LiquiLift Trace Kit
- IV Novice Pack
- Thermoject
- 0.9% Sodium Chloride IV Flush
- IV Infusion CPI
- Monoject Sodium Chloride Flush Syringe
- Monoject Prefill Sodium Chloride
- Active Injection Kit LM-Dep-1
- Active Injection Kit LM-DEP
- Dyural-80
- Dyural-40
- Tip-Lok Diluent
Overview of Research Findings
The Burkholderia cepacia (B. cepacia) complex, encompassing approximately 24 closely related bacterial species, exhibits a versatile metabolism that facilitates rapid adaptation and mutation. This adaptability contributes to its resistance to antiseptics and antibiotics, as well as its ability to thrive in environments with limited nutrients. Such characteristics elevate the concern of B. cepacia contamination in medical devices and liquid pharmaceutical products, leading to significant consequences and prompting recalls of IV flush syringes.
Resistant to standard disinfectants, B. cepacia poses a substantial threat, particularly in healthcare settings where its opportunistic nature becomes apparent. The bacteria, responsible for deadly infection outbreaks linked to IV flush syringe recalls, disproportionately affects individuals with compromised immune systems, such as those with pre-existing conditions like cystic fibrosis or HIV-positive patients, chemotherapy recipients, and the elderly.
The adaptability of B. cepacia to stressful environments, including water bodies and liquids with minimal nutrients, presents challenges in targeting and eradicating it. Notably, the bacteria can survive and multiply in the saline solution found in IV flush syringes, contributing to widespread contamination and infection outbreaks.
The absence of an instant test for identifying and detecting B. cepacia further complicates the situation. In healthcare settings, where timely responses are crucial, the bacteria’s swift spread and multiplication heighten the risk for vulnerable patients, leading to severe bloodstream infections. This alarming scenario underscores the importance of addressing contaminated syringes, especially those related to prefilled saline flush syringes, heparin IV flush syringes, and Nurse Assist saline flush, to prevent the occurrence of Burkholderia cepacia bloodstream infections associated with intravenous lines.
Recalls
Over the years, several manufacturers voluntarily recalled defective IV flush syringes for different reasons.
Nurse Assist Inc. announced a voluntary recall of several batches of unexpired IV Flush Syringes on 4th October 2016. The company distributed the recalled syringes between 16th February and 30th September 2016.
The recalled products included:
| Lot Number | Product Code | Description |
| All unexpired lots | 1203 | IV Flush Syringe, 3ml |
| All unexpired lots | 1205 | IV Flush Syringe, 5ml |
| All unexpired lots | 1210 | IV Flush Syringe, 10ml |
| All unexpired lots | 1210-BP | IV Flush Syringe, 10ml |
Product Numbers 1210, 1205, and 1203 contained 180 syringes, with six inner cartons, each holding 30 IV flush syringes. Product Number 1210-BP had 400 needles in every case.
The FDA classified the Normal Saline IV Flush Syringe recall as a Class I recall because of the high risk of injury and death. The recall involved 400,000 IV flush syringe units contaminated with the deadly B. cepacia.
BD Medical Flush IV Syringe Recall
BD Medical voluntarily recalled particular BD Pre-Filled Saline Flush syringes and BD PosiFlush Heparin Lock Flush in April 2018.
This move followed concerns that the syringes had S. marcescens bacteria. The CDC discovered the link between BD flush products and severe bloodstream infections in several states.
The recalled products included:
BD PosiFlush Heparin Lock Flush Syringes -Catalog REF Numbers
- 306509
- 306510
- 306511
- 306512
- 306513
- 306514
- 306515
- 306516
- 306517
- 306521
- 306525
- 306528
- 306531
BD Pre-Filled Saline Syringes – Catalog REF Numbers
- 306500
- 306502
- 306503
- 306504
- 306505
- 306507
- 306508
- 306518
Other notable IV flush syringe recalls included:
- February 2018
Due to improper packaging, Medline Industries Inc. recalled 90,870 USP 5 mL in 10 mL Syringe ZR syringes.
- August 2015
Covidien LLC recalled 207,876 Monoject 0.9% Saline Flush syringes due to compromised sterility during the packaging process.
- April 2015
MRP, LLC dba AMUSA recalled 200,000 USP Flush Syringes due to wrong expiration dates indicated on the label.
- July 2014
Becton Dickinson & Company recalled 3,088,320 PosiFlush SF Saline Flush syringes due to open seals on some devices.
- August 2013
Covidien LLC recalled 3,565,980 Monoject 100 Units/mL Heparin Flush syringes due to non-sterile water in the needles.
- November 2011
Hospira Inc recalled 5,817,600 USP pre-filled flush syringes due to contamination in the saline fluid.
- November 2010
Covidien LP recalled 1,356,480 MONOJECT Prefill Heparin Flush Syringes due to contamination. The heparin in the syringes was oversulfated, which triggered severe allergic reactions in some people.
- September 2010
Excelsior Medical Corp. recalled 13,734,640 Excelsior Disposable syringes due to possible leakage and compromised sterility.
- July 2007
B. Braun Medical Inc. recalled 1,325,760 Normal Saline syringes due to contamination.
- October 2006
Excelsior Medical Corp. recalled 466,000 Sodium Chloride Flush syringes due to improper labeling.
General Physician Sentiment
Bacteremia infections often trigger life-threatening reactions in the body. One of the most significant concerns is that the condition can advance to septicemia.
Sepsis can be challenging to identify and treat. If left untreated, it can cause organ failure, extensive tissue damage, and death in some cases.
According to the National Institute of General Medical Science, more than 1.7 million people in the US suffer from sepsis yearly, and 28% to 50% of these cases result in fatalities.
In 2011, the US Agency for Healthcare Research and Quality (AHRQ) ranked septicemia as the most expensive medical condition. The hospital costs related to septicemia were over $20 billion that year.
Dr. Amesh Adalja, an infectious disease expert at the Johns Hopkins Center for Health Security, explains why the B. cepacia and S. marcescens are so severe.
He cautions that the main reason the bacteria are so dangerous is that they get injected directly into the bloodstream. There are no barriers that limit entry into the bloodstream.
Most patients reported symptoms such as:
- Elevated heart rate
- Disorientation or confusion
- Shortness of breath
- Sweaty or clammy skin
- Shivering or chills
- Fever
Brief Lawsuit History
IV Flush Syringe Lawsuits are currently in the early stages, with no notable settlements thus far. Legal experts remain optimistic about positive outcomes, anticipating that manufacturers involved in the lawsuits might opt for out-of-court settlements to avoid the demanding court processes. If you or your loved one experienced a blood infection resulting from contaminated syringes, pursuing an IV Flush Syringe Lawsuit is crucial.
Seeking compensation for injuries caused by antibiotic-resistant bacteria, saline flushes, pre-filled syringes, administering multiple medications, clear plastic syringe use, disease control issues, bone marrow transplant complications, and symptoms such as sweaty skin and confusion is the best way to hold manufacturers accountable.
B. Braun Medical Group Lawsuit settlement
Although AM2PAT produced the defective syringes, B. Braun branded and sold the products. This makes B. Braun liable for the harm the contaminated needles caused.
The company agreed to settle the criminal charges filed by the government by paying $7.8 million. $4.8 million was forfeited profit and penalties, while $3 million went to restitution for people affected by the contaminated syringes.
It’s Time for Negligent Manufacturers to Be Responsible; Join the Many
Join the Many empower victims harmed by negligent companies’ products. We believe the justice system exists for people like you and your loved ones, not big businesses that refuse to take accountability.

