In 2016, the CDC noted and monitored a Burkholderia Cepacia outbreak related to IV flush syringes. The CDC’s move followed a growing number of bloodstream or bacteremia infections across five states. In January of the following year, the CDC launched an investigation into about 162 cases involving B. cepacia infection and contaminated Nurse Assist Saline Flush IV syringes. Seven out of these cases were fatal. The manufacturers finally recalled the bacteria-contaminated flush syringes. Unfortunately, dozens of people still suffer from severe bloodstream infections.
If you or your loved one developed sepsis or bacteremia infection due to a contaminated IV Flush syringe, Join the Many who share your experience. Together, we can hold device makers accountable and recover what you lost.
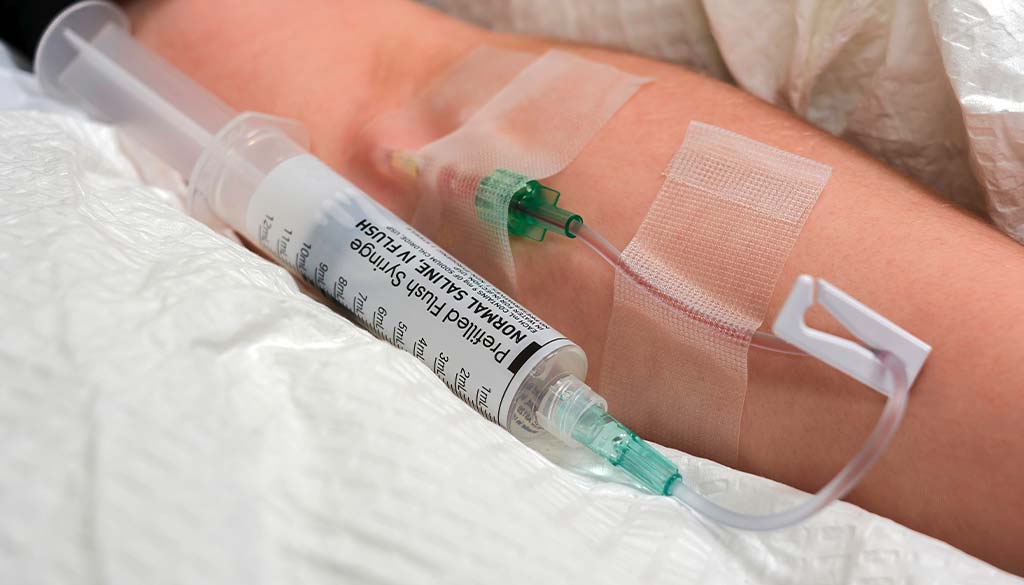
Brief History IV Flush Syringes
In a healthcare setting, nurses flush or clear a patient’s IV line by injecting a colorless liquid in a clear plastic syringe. Flushing the IV helps to ensure the medicine reaches the bloodstream by keeping the line clear of any blockage.
Most pre-filled IV flush syringes contain normal saline containing water and 0.9% sodium chloride or salt. Normal saline is compatible with the chemical processes in the body. It helps to ensure proper fluid balance because it’s the closest match to the regular salt concentration in the blood. Some IV flush syringes contain heparin, an anticoagulant, instead of pure saline. Heparin IV flush syringes have different concentrations of heparin diluted in standard saline. Heparin stops clots from developing inside the IV line or catheter. However, not everyone should use these heparin IV flush syringes. Heparin can trigger severe reactions in people with low platelet counts or uncontrolled bleeding.
Over the years, manufacturers had to recall contaminated IV flush syringes. One of the most outstanding recalls occurred in 2016. B.D. Medical and Nurse Assist Inc. recalled multiple batches of needles following an outbreak of B. cepacia infections in various states.
The same year, state and federal agencies collaborated with the CDC to monitor the infection outbreak. Most IV flush syringes involved in the investigation were products of Nurse Assist, a Texas-based company. The outbreak caught the CDC’s attention after a group of patients from a nursing home in Maryland reported B. cepacia infections.
By October 2016, five state health departments had confirmed multiple B. cepacia infections linked to Nurse Assist I.V. Flush Syringes.
By January 2017, the number of cases had more than doubled to 164 in about 59 different facilities across the five states.
The affected states included:
- Pennsylvania
In Pennsylvania, health officials confirmed syringes had B. cepacia and 30 cases of infections. The cases involved Nurse Assist Products and occurred in ten different facilities.
Deadly bacteria in the contaminated IV flush syringes caused at least two fatalities since January 2017.
- Maryland
Health officials in Maryland identified 12 B. cepacia infection cases during the same period. The cases involved Nurse Assist pre-filled syringes and occurred in three different facilities.
- New Jersey
Health officials in New Jersey also confirmed 59 B. cepacia infection cases in 20 different facilities. The patients were in long-term care facilities where nurses administered drugs via central IV lines.
In September 2017, New Jersey officials cautioned about 43 facilities about the contaminated IV flush syringes.
- New York
The State Department of Health in New York confirmed about 58 B. cepacia bloodstream infections cases. The cases occurred in 24 facilities involving contaminated Nurse Assist flush IV syringes.
The department cautioned more than 50 long-term care facilities about the possible contamination. There were five confirmed cases of fatalities caused by the deadly infections.
- Delaware
In Delaware, health officials identified four cases of the deadly B. cepacia infections. The four cases occurred in two different facilities. In October 2016, Nurse Assist Inc. finally voluntarily recalled the Saline Flush IV Syringes linked to B. cepacia infections.
The voluntary recall involved about 386,175 IV flush syringes issued across the country. Nurse Assist, Inc. distributed the recalled batches from February to September 2016.
The FDA categorized this voluntary recall under Class I recalls, which are the most serious recalls. According to the agency, a Class I recall implies that medical devices could cause death or severe injuries.
Serratia Marcescens and B. Braun Heparin IV Flush Syringe
Nurse Assist Inc. wasn’t the only manufacturer involved with the contaminated IV flush syringes. In 2016, the federal government filed a lawsuit against B. Braun Medical Inc. for distributing contaminated pre-filled saline IV syringes in 2007.
The Department of Justice (DoJ) stated that another company manufactured the saline flush syringes. But, the contaminated needles carried the B. Braun label.
B. Braun had an agreement with the company AM2PAT to manufacture the syringes and started buying them in March 2006.
Even before entering the agreement, B. Braun knew about some manufacturing issues at AM2PAT facilities. To address the problems, AM2PAT informed B. Braun of a plan to relocate the manufacturing facility in the spring of 2007.
AM2PAT also intended to contract a different company to ensure better sterilization of the B. Braun saline IV syringes. The new company would utilize a new radiation sterilization procedure.
However, the company’s quality department did not sanction the changes. B. Braun started distributing the new saline syringes from AM2PAT’s new facility before approval. The department still needed to approve the new sterilization method and company.
Shortly after B. Braun started distribution, they got complaints about the new syringes altering color. AM2PAT claimed there was a plan to adjust the radiation process but did not flag the changes.
About two months after the new syringes from AM2PAT hit the market, B. Braun recalled all the batches. The main reason was that the new radiation sterilization caused deadly white particles to form in the saline inside the syringes.
After the voluntary recall, AM2PAT admitted to sharing incorrect information with B. Braun regarding the new radiation sterilization procedure.
Despite having the information, B. Braun continued getting the saline syringes from AM2PAT. Before a month ended after resuming sales, it became clear that the saline syringes had bacterial contamination.
The Serratia marcescens bacteria found in the syringes can trigger severe blood infections. The contaminated needles caused blood infections in about 100 patients from Nebraska, New York, Texas, and California.
Unfortunately, five of the Serratia marcescens blood infection cases were fatal. Among the affected patients was a young man from South Florida, Kyle Pacheco, who was nineteen years old.
Kyle underwent a bone marrow transplant and was in recovery when he used a contaminated syringe. As ProPublica highlighted, Kyle developed a severe infection that caused him to remain in a coma for one month. He could not walk after waking up and eventually lost his life in 2010.
After the discovery, B. Braun issued a recall for the affected syringes. However, the damage was done, and dozens of vulnerable people were at risk of deadly blood infections.
Overview of Research Findings
The October 2016 Nurse Assist Normal Saline IV Flush recall followed serious concerns in the medical community. This deadly bacterium consists of 18 different species of bacteria.
These species trigger pneumonia in people with conditions that compromise the immune system. The bacteria also increase the severity risk in people with underlying sickle cell anemia and lung disease.
Since no instant test can test for B. cepacia, the only way to confirm the infection is by extracting a culture from the patient’s sputum or blood. In Europe and North America, B. cepacia bloodstream infections rank seventh as the leading cause of death.
In 2015, a team of researchers evaluated the long-term mortality and leading causes of death in patients suffering from bloodstream infections. The study investigated data spanning twelve years.
The team discovered a link between bacteremia and a higher risk of mortality. The risk of death remained high for up to ten years after treating the initial infection.
What Is Burkholderia Cepacia?
Burkholderia cepacia complex is a bacteria family typically resistant to standard antibiotics.
The name originated from the person who discovered the family of bacteria, Walter Burkholder. Previously, the bacteria were identified as Pseudomonas cepacia.
Initially, agricultural-based companies developed B. cepacia for commercial purposes. It was helpful as an antifungal agent that protected forests and crops.
Environmental-based companies also discovered potential use for B. cepacia to manage pollution. The bacteria could potentially degrade various pollutants in the water and soil.
In the ’90s, the Environmental Protection Agency (EPA) registered some bio-control products. These products contained various strains of B. cepacia and had different uses.
However, the agency withdrew the registration of the bio-control products after risk assessments. They also suspended the bio-control agents with B. cepacia based on the threats the products posed to public health.
Although B. cepacia doesn’t pose risks for healthy people, it can trigger various issues in compromised patients.
In 2011, a South Korean study discovered that B. cepacia bacteremia infections increased the mortality rate in ICU patients. The mortality rate within 28 days was about 41%.
In most cases, B. cepacia penetrates the body via the respiratory tract. Since B. cepacia is often resistant to standard antibiotics, the CDC advises doctors to create treatment plans based on the particular patient.
Bacteremia Bloodstream Infections
If the initial infection remains untreated, it can quickly spread throughout the body, causing life-threatening conditions.
According to the CDC, the most common symptoms of B. cepacia bloodstream infections include:
- Shortness of breath
- Elevated heart rate
- Fever
- Sweaty or clammy skin
- Shivering or chills
- Disorientation or confusion
Bacteremia can also trigger sepsis, a potentially life-threatening reaction of the body to an infection.
Sepsis is often challenging to diagnose and requires intense treatment. It often causes multiple organ failure, extensive tissue damage, and death in some cases.
Sepsis can also cause:
- Chronic obstructive pulmonary disease
- Heart failure
- Pneumonia
- Acute heart attacks
If you or your loved one developed severe blood infections or sepsis from a contaminated pre-filled IV flush syringe, you should pursue compensation for your injuries.
Lawsuit History
The consequences of contaminated IV Flush Syringes are so severe, and the side effects of infection can last up to a decade. The manufacturers should take responsibility for the injuries the devices cause to vulnerable people.
IV Flush Syringe Lawsuits are still proceeding as more affected people continue to take legal action. Although there are no notable settlements for personal injury lawsuits, attorneys are gearing up for the anticipated litigation process.
If you or someone you care about suffered such profound harm, you deserve compensation for your injuries, pain, and suffering. You can take a bold stand and file an IV Flush Syringe Lawsuit by filing a claim. The first step is contacting Join the Many.
IV Flush Syringe $7.8 Million settlement
The government investigation into B. Braun’s contaminated IV flush syringes highlighted several issues. B. Braun wasn’t the original manufacturer, but they knew AM2PAT Inc.’s history of deficiencies.
AM2PAT had a long history of manufacturing issues, and the FDA had already issued the company several warning letters. Despite this knowledge, B. Braun proceeded to acquire and resell the defective AM2PAT syringes.
As the DOJ explained, AM2PAT used improper manufacturing processes that produced contaminated syringes. The company’s list of failures included the following:
- Using filthy equipment to make syringes
- Failing to offer adequate employee training
- Failing to run tests on the equipment to ensure proper function
- Failing to maintain clean environments in the factory and around manufacturing equipment
AM2PAT later permanently ceased operation and let go of the entire staff. In May 2016, B. Braun agreed to settle penalties and forfeiture worth $4.8 million.
The company also paid an additional $3 million in restitution. The amount covered the criminal liability for distributing the contaminated syringes. This significant settlement indicates there’s a potential success with personal injury lawsuits.
Are You Ready to Take Action? Join the Many
Join the Many empowers people like you who suffered injuries due to the products of negligent corporations. We can educate you about your legal options and help you navigate the complex legal system, making the entire process of recovering your losses much easier than doing it on your own.