Did you have an IVC Filter placed? Did you then suffer numerous health complications, perhaps to your heart, lungs, or other organs resulting from this filter?
Vena cava filters, IVC filters, are a type of device placed into the vena cava vein that runs through the center of the body. They are supposed to work to prevent a blood clot from traveling through a vein to the lungs or heart, a condition called a pulmonary embolism. However, IVC filters have complications, some of which are very complicated and life-threatening. Many people have pursued financial compensation because of the losses suffered by the use of these products.
We can help determine if you are owed compensation for the losses you’ve incurred as a result of having an IVC filter placed, then fight alongside you to hold device manufacturers accountable.
What Is an IVC Filter?
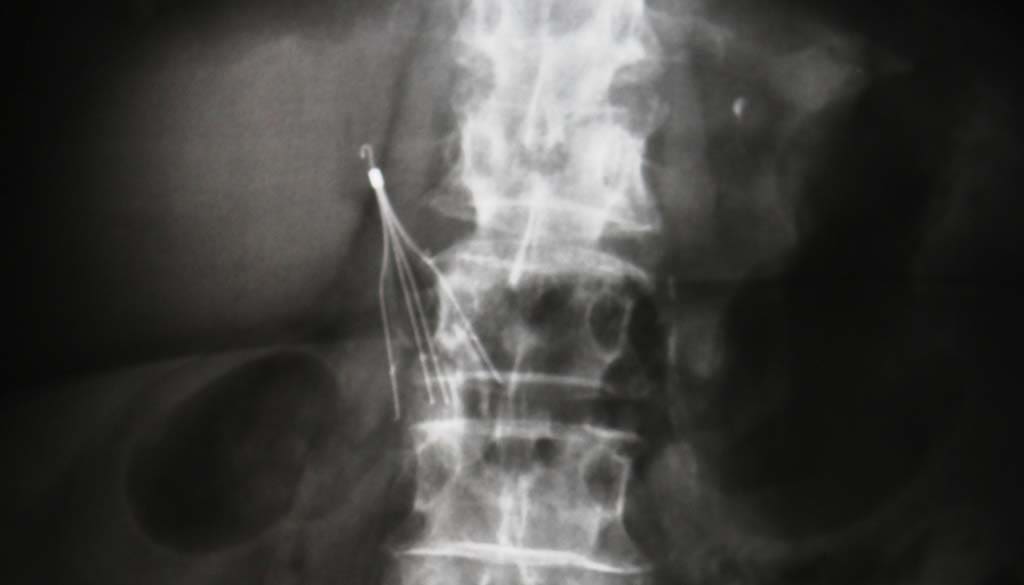
The Cleveland Clinic provides insight into what the IVC filter is and how it works. The filter is designed to prevent a pulmonary embolism from occurring. These may be a necessary tool for preventing complications from blood clots in some people, especially those who cannot take blood thinners.
In some cases, the filters are permanent, which means they remain in place long term. Others are retrievable, meaning they can be placed for a short time and then removed after the risk is lower.
The IVC filter is placed through a small incision made in the neck or groin area. A catheter, which is a thin tube, is placed into this vein and threaded up to reach the vena cava vein. There, the doctor will position the filter and expand it, allowing for the device to remain in place, holding onto the sides of the vein’s walls.
Who May Need an IVC Filter?
IVC filters are used for a variety of situations when there are risks of blood clots forming or when blood clots already exist. This treatment may be beneficial to people who:
- Are unable to use blood thinning medications known as anticoagulants. This could be due to a high risk of bleeding or negative reactions to them.
- Have had episodes of venous thromboembolism, or VTE, in the past, even when they were taking blood thinning medications.
- Have had a deep vein thrombosis, DVT, or at high risk for them
- Have or had a blood clot that made its way to the lung, causing a pulmonary embolism, and could suffer the same complications again.
Some people who are pregnant, have suffered a serious injury, those who have blood clotting disorders, and those who have had restricted movement due to trauma or surgery may have these devices recommended for them.
According to information from the Cleveland Clinic, there have been more than 250,000 filters placed in the U.S. since 2012. In addition, that year, surgeons inserted more than 25 times the number of IVC filters in the U.S. as were placed in the five largest countries in Europe.
What Are the Known Risks of IVC Filters?
Since their initial approval and start of use, there have been numerous changes to the known risks and side effects of using IVC filters. Some of the most common and noted risks, according to John Hopkins Medical Center, include:
- Allergic reactions
- Infection
- Excessive bleeding
- Damage to the blood vessel at the location where it is being inserted
- Blockage of the blood flow through the vena cava, causing leg swelling
- Filters can pierce through the inferior vena cava, causing pain and damage to organs
- The filter can move from one area to the next, including to the heart and lungs, causing injury as well as death
- Difficulties with placement of the filters is possible
- There is still the risk that a blood clot can continue to travel to the lungs
The History of the IVC Filter
The IVC filter has been used for decades after having been developed in 1969. Its initial development was by Kazi Mobin-Uddin, MD, who published his findings in the New England Journal of Medicine. Over time, this product was updated to the Greenfield filter, which was said to have a lower risk of complications, according to the National Library of Medicine.
The first IVC filters were approved by the U.S. Food and Drug Administration (FDA) in 2002. At that time, the C. R. Bard devices were designed and called Recovery IVC filters. In 2003, the FDA approved the Tulip IVC filter from Cook Medical.
After receiving complaints about adverse complications to these products, Bard decides to pull the Recovery IVC filter off the market. It replaced it in 2003 with the G2 IVC filter.
All Names Used for IVC Filters
These products are manufactured by a number of companies, and there have been several versions of them. Some of these include:
- C.R. Bard with Recovery and G2 Express products
- Boston Scientific with Greenfield products
- Cook Medical
Overview of Research on IVC Filters
A number of studies have been released over the last few years that have shown significant concerns with the use of these products. Here is a look at some of those.
In 2010, a JAMA Internal Medicine study released findings that a high number of people who had Bard IVC filters placed suffered serious complications from them. The study found that 13 of the 80 patients in the study had at least one fracture of the device, accounting for 16% of users. In addition to this, 1 in 7 of the 28 tested devices led to the filter fracturing and causing an embolism. In 5 of 7 of those cases, the patient experienced the fragment from the IVC filter making its way to the heart – representing 71% of cases.
In three patients, ventricular tachycardia and tamponade occurred. One patient died as a result of the device’s migration to the heart. The study found that the Bard G2 IVC filters created a very high risk of failure rates and life-threatening complications in those who used them.
At this point, the U.S. Food and Drug Administration (FDA) made the decision to issue a safety communication for the device. At this point, it had received 921 filter related adverse event claims – in just a span of five years. These risks included:
- Fracture of the filter
- Migration of components to other areas of the body
- Perforation of the inferior vena cava from the device
- Embolisms
In this FDA safety notification, it recommended that the device be removed when the threat of embolisms was lower to prevent the onset of these high risks.
Studies continued to provide evidence of key concerns:
- The devices were placed in people, such as in trauma events, after the highest risk of bleeding had passed, making them not the ideal solution, and instead, the use of anticoagulants should have been used.
- Though the FDA warned that the devices should be removed between 29 and 54 days after being implanted, many were not removed. This allowed them to remain in patients longer, increasing the risk for complications.
An NCB study and report in 2015 found that 27 people had died and hundreds of additional people had suffered complications related to the use of the Bard G2 IVC filters. The report found that the FDA approved the use of the G2 series just four months after its application for the product.
The company was found to have known that these devices had problems, but instead of recalling the use of them, they instead pulled the Recovery line and replaced it with the G2 line.
Numerous additional studies have been done to show the risks of using these devices. There is also a high percentage of people who may not have needed them.
Do these complications sound like ones you’ve had? If so, Join the Many may be able to help you obtain the financial compensation owed to you as a result of your losses.
Recalls on IVC Filters
IVC filters have been recalled by the FDA over the last few years. Here is a look at a few of those cases:
Cordis OptEase – In April of 2013, Cordis Corporation recalled the use of the OptEase IVC retrievable filter. This was done because they believed the implantation instructions were not clear and the device could be inserted backward. In this situation, 33,000 devices were recalled after being placed between 2010 and 2013. The FDA issued a Class 1 recall, the highest level, noting it could cause injury or death.
Greenfield IVC Filter – Greenfield IVC filters were called twice. In August of 2005, the recall was issued as a Class 2 recall. It noted that the lack of taper on the sheath of the developed system created a higher risk of catching on a vein and tearing it. This recall of the product by manufacturer Boston Scientific stated a person could suffer temporary or medically reversible health complications. In December of 2005, a second recall, this time a Class 1 was issued for the device noting the device could detach and allow a clot to pass through it.
Lawsuit History on IVC Filters
The first cases against IVC filter manufacturers began in 2012. At that time, many people came forward to state that they believed the manufacturers of these products failed to create a safe product, failed to warn about the risks, and caused a variety of health complications, including a high risk of debt.
There are many cases that are still pending throughout the U.S. In October of 2014, the U.S. Judicial Panel on Multidistrict Litigation (MDL) ordered that the cases be consolidated into a single case in Indiana. This could help to streamline the efforts to determine if there is merit to the lawsuits.
Bard and Cooks Medical initially won some of their first cases brought against them for IVC filters. They did so because of the passing of the statute of limitations or other reasons. However, these initial cases were not the end of the story.
In fact, many people have gone on to win millions of dollars in claims after juries found the manufacturers responsible for their losses. Here are a few examples:
- In Texas, a person was awarded $1.2 million when the IVC filter dislodged and caused pain and industries, including perforation of the blood vessels and other organs.
- In March 2018, a woman was awarded $2.6 million after a filter fractured and then migrated out of place, creating complications after perforating the IVC.
- In February 2019, a jury awarded a $3 million settlement after a filter deteriorated in a Georgia woman’s body.
There are still thousands of pending IVC filter lawsuits across the U.S. It is likely that a class action lawsuit could form. However, individuals who have had these types of losses need to take action to protect their rights to compensation.
Join the Many is ready to help. If you or your family were impacted by these devices, there’s no time to waste. Contact us to learn if you may qualify for a settlement. We’ll help you understand how to exercise your legal rights and benefit from holding manufacturers accountable. There are no legal fees unless you win compensation.

