If you were one of the many people harmed by Essure, Join the Many who want to hold device manufacturers accountable. We can answer your questions and determine if you may qualify for a settlement. The legal system is confusing and frustrating, but we’re committed to making your fight as stress-free as possible by doing most of the heavy lifting for you, including matching you with the best legal care for your situation.
What Is Essure?
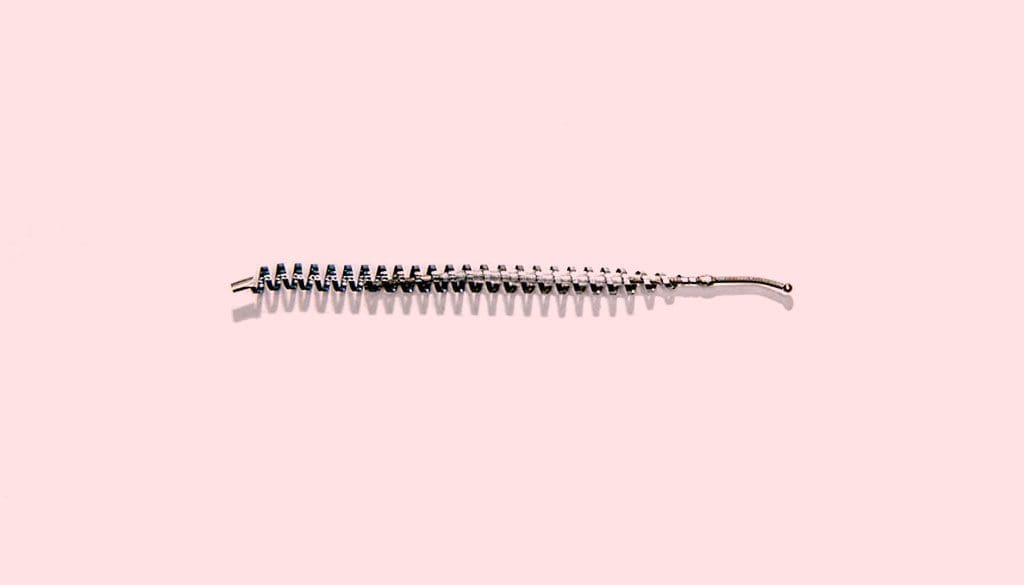
Essure is a type of sterilization device with a metal coil that’s placed into each of the fallopian tubes. This creates a blockage and fibrosis to occur. The product was initially designed by the company to create an alternative to more invasive tubal ligation. This product was designed to be put into place and to remain present for a person’s entire lifetime. However, it was only approved in the U.S. for short term use initially. According to the New York Times, more than 75,000 women had these devices placed throughout the world.
The product, developed by Conceptus Inc., was first used in the U.S. in 2002. Bayer AG of Germany then acquired the company in 2013. The product was used for many years without any recognition of the problems associated with it. However, the device was found to cause implantation problems, and that led to adverse effects, including perforation of the uterus, persistent pain, allergic or hypersensitivity reactions, and additional complications.
Research Findings and Complications with Essure Use
The Food and Drug Administration (FDA) in the U.S. is working to support the recall and other needs of patients who used Essure throughout its use period. There have been many complaints brought against the company due to the complexity of its function and its inferior use. Though this product is no longer used in the U.S., its complications may continue to impact many women. For that reason, the FDA will continue to monitor the Essure data to ensure proper support for patients is made available.
On December 31, 2018, Bayer made the decision to stop selling Essure devices and no longer distributed them in the U.S. However, healthcare providers had the ability to continue to implant these devices beyond that date – up to one year after the time they purchased the device. As a result, many people received these devices after Bayer stopped distributing them.
If you used Essure at any time, you could be the victim of a medical device malfunction. Join the Many may be able to help you to determine if you have a case. We can also connect you to attorneys who are already working on lawsuits related to Essure.
In September 2019, Essure sent a notice to all of its customers that these units that have not been implanted should be turned into the company by the end of 2019. Therefore, as of December 2019, all unused devices should have been returned and, as such, should not have been implanted beyond that time.
The FDA states that women who currently have the device and do not have complications from its use can continue to use it. They believe it is still effective at preventing pregnancy.
Bayer recognizes that there are problems with the device. The company states that there have been some patients that have reported adverse effects from the use of the Essure System. Some of those risks include the following:
- Perforation of the uterus
- Perforation of the fallopian tubes
- Devices that are located in the pelvic or abdominal cavity
- Chronic pain
- Allergic or hypersensitivity reactions
What Actions Did the FDA Take?
Here’s the breakdown of what occurred with the FDA and its use of Essure products.
In 2002, the FDA approved the use of Essure as a Class III medical device. That allowed Conceptus and later Bayer to begin using the product in patients. The companies were required to provide the FDA with any updates on efficacy and safety risks patients were experiencing while using the device.
In February 2016, the FDA required that Bayer complete a new clinical trial for Essure. The goal of this trial was to determine if there were any risks associated with the use of the device for some women. This came after there were numerous reports filed by women to the FDA because of complications they experienced using the device.
The FDA continued to receive complaints about the device. As a result, in November 2016, the organization issued a black box warning for the use of Essure devices. This is a label for the product that provides information to the public about the risks associated with the use of Essure. This step required Essure devices to have a label that warned specifically about abdominal pain, perforation risks, and allergic risks. This did not stop the product from being sold but required that patients be informed about the risks.
In April 2018, the FDA continued to receive complaints about the product’s use and effectiveness. At this point, more than 26,700 complaints about the product had been filed with the FDA directly. That spans from 2002 (since the initial launching of the product) through 2017. As a result of this, the FDA issued a restriction on sales of the device. That meant that, though some people could still obtain it, the FDA was no longer recommending its use.
In December of that year, Bayer issued the decision to stop selling the product. At the same time, the FDA took action and created a post-market surveillance plan for the product. That means that the FDA would continue to follow what people were reporting and take action as needed beyond that point. Remember, thousands of these devices were still being used with consistency. This step was a necessary one since the company removed the product from the market.
Lawsuit History and Actions
Many people suffered complications to their health resulting from their use of Essure. As a result of this, many people have sought financial compensation from Essure’s manufacturer Bayer, stating, for example, that the company did not provide a safety device or did not provide necessary warnings of risks of complications.
According to documentation from The Washington Post, Essure was promised as a product with a 99.74 percent success rate and labeled as the most effective form of permanent birth control available. In the description of the product to consumers, Essure is said to have two “soft and flexible inserts” and that the product requires a “gentle, non-surgical” procedure. For many patients, this was misleading.
Data from clinical trials completed by Bayer show that it has a device insertion failure rate between 4 and 12 percent of the time. However, there is little information to confirm this.
What Patients Experienced
The lawsuits in these cases have been brought for many reasons and due to various losses women have suffered as a result of their Essure use. This includes health complications, hospitalizations, and ongoing pain. About 9,000 women had to have the device removed. To do this, it requires a surgical procedure that, in many cases, requires a full hysterectomy.
Because of the sheer number of reported complications – which was as high as 33,000 recently at both the state and federal court levels – a Multidistrict litigation panel (MDL) was created for the Essure clients. This was done in 2016 as a result of 28 people who had submitted a petition urging it. However, 23 of the 28 cases in that grouping were in Pennsylvania. As a result, a judge in the Eastern District of Pennsylvania motioned for the MDL to be withdrawn and instead agreed to hear the case in the state.
In all, over 10,000 injuries were reported to the FDA. This includes 30 deaths that are believed to have stemmed from the device. Of those, 4 deaths occurred in adult women, and 21 claims were for pregnancy loss. In two additional cases, the infants died after their birth as a result of the device’s use.
It’s important to note that there are many other lawsuits across the country. Each state with complaints establishes its own process of managing those claims. There are numerous states where people have decided to take action.
Outcomes from the Cases
There are many cases still pending, and a case action lawsuit may occur as well. It is noted that in November of 2016, Bayer Healthcare is said to have paid out $413 million in legal-related compensation, though the official amount awarded has not been clearly outlined. In 2019, they reported 33,000 pending lawsuits.
It is likely that additional settlements will occur over time. There is no way to know how much they may be just yet. However, anyone that has suffered losses due to the use of Essure should request help from Join the Many now. There is no cost to you and no risk, but you may be entitled to financial compensation for the losses you’ve incurred as a result of these products.
Lawsuits in the case are numerous with various claims, including:
- The company promoted the product inaccurately, stating that the process was a 30 minute non-surgical and simple procedure.
- Essure failed to get full approval and complete necessary safety studies before using the product on women
- The product injuries were not reported and acted upon soon enough
- There was no warning to women about the risks associated with the device or the removal of the device
- Some women still got pregnant even with the device in place, even with a very high promised success rate.
- Death occurs as a result of the device’s use in both unborn children, children born who later died, and adult women who used the device.
It is also important to note that when Bayer removed the product from the market, it did not state it was doing so because of these claims or because of any fault in its product. They stated it was due to declining sales of Essure.
There was no formal recall completed for Essure. The FDA did not provide any specific recall requirements to the company.
Essure’s manufacturer has reached a settlement with many claimants, though they have not admitted any guilt in the process.
What You Can Do Now
If you used Essure, or you are continuing to use it, and the device failed you or caused complications to your health, you should take action immediately. The first step is to work with your doctor to determine the best type of treatment for your needs. In some women, this has included the removal of the device.
In addition to this, you may be owed for the suffering you’ve endured because of Essure. This may include losses related to medical bills as well as pain and suffering. You may also be entitled to compensation if your loved one, such as an immediate family member, died as a result of complications from Essure products.